|
Bamford-Stevens Reaction. Tosylhydrazones give alkenes upon treatment with strong bases. This reaction is performed in two steps, where the intermediate diazo compound may be isolated. Subsequent reaction with protic or aprotic solvents strongly influences the outcome of the reaction. This reaction may be used to effect. The Bamford–Stevens reaction is a chemical reaction whereby treatment of tosylhydrazones. (also in the Collective Volume (1990) 7:438 (PDF)); ^ Sarkar, T. 'Silicon-directed Bamford-Stevens reaction of β-Trimethylsilyl.

  Media in category 'Bamford-Stevens reaction'
The following 22 files are in this category, out of 22 total.
Retrieved from 'https://commons.wikimedia.org/w/index.php?title=Category:Bamford-Stevens_reaction&oldid=309684075'
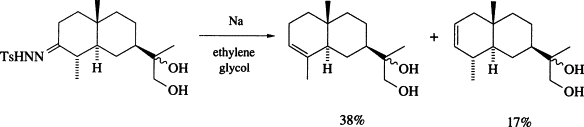
Decomposition of tosylhydrazones of carbonyls in a basic medium is one of the most important methods for synthesis of diazocompounds (Bamford-Stevens reaction)1. This reaction with arylaldehyde, arylalkyl- and diaryl ketone tosylhydrazones is usually carried out in the presence of sodium alkoxides in alcohols, glycols, their ethers or in pyridine. Diazocompounds stabilized by electron withdrawing groups can be generated under milder conditions (diluted aq NaOH solutions or basic Al2O3). The standard conditions of the Bamford-Stevens reaction are not suitable for the synthesis of diazoalkanes derived from aliphatic aldehydes and ketones.
 Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2022
Categories |
|||||||||||
 RSS Feed
RSS Feed
